Efficacy and Safety of a Patient-positioning Device (EZ-FIX® Cushion) during Endoscopic Submucosal Dissection of Gastric Epithelial Neoplasm: A Prospective Randomized Trial
Article information
Abstract
Background/Aims
Cooperation of patients plays an essential role during gastric endoscopic submucosal dissection (ESD) for successful outcomes. We aimed to assess the efficacy and safety of a patient-positioning device (EZ-FIX®) during ESD for gastric epithelial neoplasm.
Materials and Methods
In this prospective study, 86 consecutive patients with gastric epithelial neoplasm scheduled for ESD at the Pusan National University Hospital were included and randomly assigned to the EZ-FIX® (n=44) or non-EZ-FIX® (n=42) groups. The primary outcomes measured were endoscopist satisfaction profiles and contribution level of EZ-FIX® to the procedure.
Results
No significant differences were observed between the two groups regarding patients’ clinicopathologic characteristics, though the mean procedure time was longer in the EZ-FIX® group (P=0.044). In the EZ-FIX® group, 16 patients (36.4%) were categorized as a contribution group. Subgroup analysis between the contribution and non-contribution groups revealed that the contribution group had a larger lesion size (P=0.043) and a longer procedure time (P=0.037) and showed a higher patient’s movement score (P<0.001) with a higher dosage of propofol (P=0.004) and pethidine (P=0.001) required. Endoscopist satisfaction scores on sedation (P<0.001) and overall procedure (P=0.010) were lower in the contribution group.
Conclusions
Thus, EZ-FIX® might be helpful especially for patients who are expected to exhibit uncooperative sedation or those with a large lesion size, which would necessitate a longer procedure time.
INTRODUCTION
Endoscopic submucosal dissection (ESD) has been widely accepted as an excellent treatment for gastric epithelial neoplasm [1-4]. Although achieving en bloc resection with a higher curability rate is the major advantage of ESD over conventional endoscopic mucosal resection [5,6], the technique of ESD is complex and requires considerable expertise [7]. Furthermore, ESD is associated with a relatively higher risk of adverse events such as bleeding or perforation in cases with a larger lesion with ulcer/fibrosis on difficult location, which needs a longer procedure time requiring at least moderate to deep level of sedation [8,9]. Aside from endoscopic techniques including the endoscopists’ experience and tumor’s characteristics, adequate level of sedation, and patients’ cooperation throughout the procedure are very important for successful outcomes of ESD. Incomplete or unstable sedation requires repetitive administration of sedative drugs and causes interruptions with subsequent lengthening of the ongoing procedure [10]. Some patients exhibit paradoxical reactions showing an abrupt or excessive movement, which can significantly impact the delay of the procedure and even discontinue the ESD procedure completely [11]. In these cases, risks of bleeding or perforation might increase [12]. However, there is no effective strategy to prevent these uncontrolled movement under conscious sedation especially for non-anesthesiologists providing sedation, and there is a paucity of data on the patient-positioning device applying for endoscopic procedure including gastric ESD.
We hypothesized that fixing the patient’s position during gastric ESD can minimize patients’ unpredictable movement, and this might lead to successful outcomes. Thus, we aimed to assess the clinical implications of EZ-FIX® (Arlico Medical Co., Seoul, Korea), a patient-positioning device using polystyrene particles and compressed air, during ESD for gastric epithelial neoplasm.
MATERIALS AND METHODS
1. Study design
This prospective randomized trial was conducted between March 2014 and June 2014 by consecutively enrolling patients with previously diagnosed gastric adenoma or early gastric cancer (EGC) who were scheduled for ESD in Pusan National University Hospital. Written informed consent was obtained from all participants prior to ESD, and each patient was randomly assigned to the EZ-FIX or non-EZ-FIX group according to a randomization table generated using Excel 2003 (Microsoft Corporation, Redmond, WA, USA). This study was approved by the Institutional Review Board of Pusan National University Hospital (1202-010-001). Our research was registered at the Clinical Trial Registration sites in Korea (http://cris.nih.go.kr) as KCT0000373.
2. Inclusion and exclusion criteria
ESD was performed for patients with gastric adenoma without any limitation in size or EGC according to the expanded criteria proposed by Gotoda et al. [13]. Exclusion criteria were as follows: 1) previous history of upper gastrointestinal surgery; 2) known hypersensitivity to midazolam (MDZ), propofol, or pethidine; 3) abnormal coagulation profile or current (within 7 days prior to the procedure) use of antiplatelet agents and anticoagulants; 4) physical status IV according to the classification put forth by the American Society of Anesthesiologists (ASA) or severe comorbidity; 5) women either pregnant or at a risk of pregnancy and lactating women; and 6) locally recurred or multiple lesions.
3. Patient-positioning device
EZ-FIX® (Arlico Medical Co.) was used as a patient-positioning device. It is filled with tiny polystyrene particles and compressed air covered with polyurethane. The steps of applying EZ-FIX® for patients were as follows (Fig. 1): 1) EZ-FIX® was placed on the procedure table in a loose shape; 2) after inflating the EZ-FIX®, the patient was placed on the EZ-FIX® in left lateral decubitus position; 3) locking all belts on the device, the patient was wrapped with EZ-FIX® including the right arm; and 4) lastly, inflated air was removed from the EZ-FIX® by using vacuum to “copy” and fix the shape of the patient’s posture. After the placement of monitoring devices, sedatives were administered.

Method of patient preparation using EZ-FIX® for gastric endoscopic submucosal dissection. (A) EZ-FIX® is filled with tiny polystyrene particles and compressed air and covered with polyurethane. (B) EZ-FIX® is placed on the procedure table in a loose shape. (C) After inflating the EZ-FIX®, the patient is placed on the EZ-FIX® in a left lateral decubitus position. (D) Locking of all belts on the device is performed by wrapping the right arm. (E) Deflation of the EZ-FIX® is performed using a vacuum which fixes the shape of the patient's posture.
4. Sedation and patient monitoring during ESD
ESD was performed by two experienced endoscopists who had conducted gastric ESD for >3 years (GHK and BEL). All medications were administered by physicians from the endoscopy division who did not participate in the actual ESD procedures. The target level of sedation was moderate to deep sedation, which is usually required for advanced endoscopic procedure, including ESD or endoscopic retrograde cholangiopancreatography (ERCP).8,14 The initial intravenous bolus of MDZ was 2.5 to 5.0 mg; a half dose was used in patients ≥70 years old or ASA III–IV patients. An additional intravenous 2.5 mg of MDZ or 10 to 20 mg of propofol bolus was injected when a patient failed to achieve proper sedation after the initial MDZ injection, and a 10 to 20 mg bolus of propofol administration was done at a minimum 1-minute interval during ESD. The initial intravenous bolus dose of pethidine was 25 mg, and an additional dose of 12.5 to 25 mg of pethidine was administered if necessary. Oxygen was supplied via a nasal cannula at a flow rate of 2 L/min, and pulse oximetry was used for intraprocedural monitoring. Blood pressure (BP) was automatically assessed at 3-minute interval and was additionally checked if necessary. After ESD completion, the total procedural time was measured from intubation to withdrawal of the endoscope and the dosing rate of propofol was calculated as total injected dose (mg) per total procedural time (min) in the ESD report.
For the evaluation of the cooperative level of the patients, the patient’s movement score during the procedure was assessed by a single assistant endoscopist and divided into four scores. None or a little movement was one. Mild or moderate movement, but did not significantly impact on the procedure, was two or three. Severe movement, leading to the interruption of the procedure or when the restraining was mandatory for the patient’s safety, was four. Since it was difficult to evaluate the own patients’ level of movement in the EZ-FIX group, the score was given on the assumption that these patients were free from EZ-FIX.
5. Outcome measurement and definitions
The primary outcomes were endoscopist satisfaction profiles and contribution level of EZ-FIX® for the procedure. The above mentioned two endoscopists completed a questionnaire, using a 10-cm visual analog scale (VAS), which assessed the level of satisfaction on sedation and the overall satisfaction score on the procedure (ranging from 0=poor to 10=excellent). In the EZ-FIX group, the contribution of EZ-FIX® toward a stable ESD procedure was also assessed and divided into four categories: none, low, medium, or high. For example, the contribution level is none for patients with no or subtle movement during ESD, while the contribution level could be none to high for patients with unpredictable big movement. The contribution group was further defined from low to high categories.
Clinical outcomes including rates of en bloc and complete resection, and procedure-related complications such as intraprocedural unstable vital signs, postprocedural bleeding, and perforation were assessed as secondary outcomes. Intraprocedural unstable vital signs were defined as oxygen saturation levels falling below 90% for ≥30 seconds, a decrease of pulse rate below 50/min for ≥30 seconds, or a decrease of systolic BP below 90 mmHg.
The patients also completed a questionnaire, using a 10-cm VAS, which assessed post-ESD myalgia (ranging from 0=no pain to 10=pain as bad as it could) and overall satisfaction on the procedure (ranging from 0=poor to 10=excellent) the day after ESD. In addition, the presence of feeling of uneasiness to the use of EZ-FIX® was investigated.
6. Sample size calculation
Taking into account the feasibility of the sample size in our institute, we aimed to conduct a pilot study to compare EZ-FIX group with non-EZ-FIX group by using 45 patients in each group. Assuming a 10% withdrawal rate, we resolved to enroll 100 patients (50 per each group).
7. Statistical analysis
Continuous variables are expressed as mean±standard deviation, while categorical variables are presented as frequencies with percentages. In the analysis, Student’s t-tests were used to compare continuous variables, and chi-square and Fisher’s exact tests were used for categorical variables. Statistical analyses were performed using SPSS version 21.0 (IBM corp., Armonk, NY, USA). A P-value of <0.05 was considered statistically significant.
RESULTS
1. Baseline characteristics
One hundred and four patients were consecutively assessed for eligibility in this prospective study, and a total of 86 patients were enrolled excluding 18 patients who had multiple or recurred lesions. Each patient was randomly assigned to the EZ-FIX (n=44) and non-EZ-FIX groups (n=42) (Fig. 2). Table 1 summarizes the baseline characteristics of both groups. While the EZ-FIX group needed significantly larger amount of propofol (total dose 104.1±82.9 mg vs. 71.0±47.3 mg, P=0.026; dosing rate 3.8±2.1 mg/min vs. 3.0±1.8 mg/min, P=0.058) and a longer procedure time (28.1±24.2 min vs. 19.6±12.0 min, P=0.044) than the non-EZ FIX group, no significant differences were observed between the two groups in terms of age, gender, body mass index, ASA classification, level of patients’ movement and tumors’ characteristics such as tumor location, macroscopic type, tumor size, and histology.
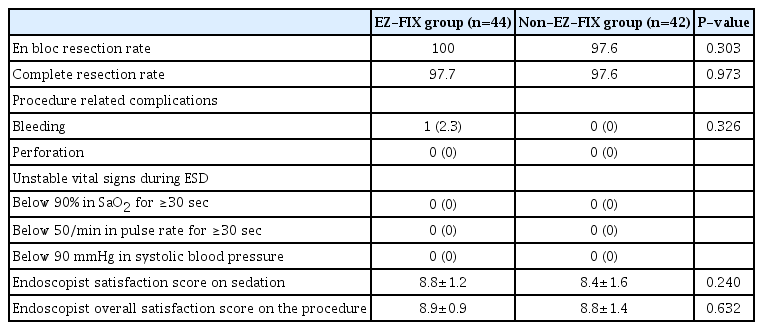
2. Clinical outcomes and endoscopist satisfaction profiles
The clinical outcomes and endoscopist satisfaction profiles according to the use of EZ-FIX® are shown in Table 2. The rates of en bloc and complete resection were not different between the two groups. Not including one patient experiencing post-ESD bleeding in the EZ-FIX group, no serious procedure-related complications occurred in both groups. Endoscopist satisfaction score on sedation was 8.8±1.2 and 8.4±1.6 in the EZ-FIX and non-EZ-FIX groups, respectively (P=0.240). The endoscopist overall satisfaction score on the procedure was 8.9±0.9 and 8.8±1.4 in the EZ-FIX and non-EZ-FIX groups, respectively (P=0.632). Consequently endoscopist satisfaction score on sedation and overall satisfaction score on the procedure did not show significant differences between the two groups.
3. Contribution level of EZ-FIX® and clinical features affecting the contribution to stable ESD
Dividing into four categories of the contribution of EZ-FIX®, 28 (63.6%) were included in none, 12 (27.3%) were in low, four (9.1%) were in medium, and no patients were in high. The contribution group from low to high categories included 16 patients (36.4%). Table 3 shows the subgroup analysis between the contribution group (n=16) and non-contribution group (n=28). The contribution group had a larger tumor size (24±19 mm vs. 15±10 mm, P=0.043) with a longer procedure time (38.0±33.9 min vs. 22.4±14.2 min, P=0.037) and showed a higher patient’s movement score (P<0.001) with a higher dosage of propofol (total dose 161.9±109.1 mg vs. 71.1±35.0 mg, P<0.001; dosing rate 4.9±2.3 mg/min vs. 3.1±1.6 mg/min, P=0.004) and pethidine (48.4±14.3 mg vs. 33.0±13.7 mg, P=0.001). Endoscopist satisfaction score on sedation (7.5±1.0 vs. 9.5±0.5, P<0.001) and the overall satisfaction score on the procedure (8.4±1.0 vs. 9.2±0.8, P=0.010) were significantly lower in the contribution group.
4. Patients’ questionnaire survey
A total of 71 patients (83.0 %) completed the questionnaires the next day after ESD, and Table 4 shows the results. No difference was observed in the overall satisfaction score on the procedure between the EZ-FIX and non-EZ-FIX groups (8.3±2.3 vs. 8.8±1.8, P=0.372). The post-ESD myalgia score was also similar in both groups (2.5±2.3 in the EZ-FIX group vs. 2.1±2.6 in the non-EZ-FIX group, P=0.494). Although four patients (10.5 %) responded to have feeling of uneasiness to EZ-FIX® use, no statistical significance was observed between the two groups.
DISCUSSION
In the present study, EZ-FIX® itself did not affect clinical outcomes of gastric ESD including endoscopist satisfaction profiles. However, we recognized that there were certain situations in which the procedure was benefited by EZ-FIX® use.
As gastric ESD requires fine, complicated techniques with a long procedure time, patients’ cooperation is essential to accomplish safe and successful outcomes [15]. Proper sedation can minimize patients’ movement, and appropriate patients’ cooperation improves the rate of en bloc resection [10] and not only reduces patients’ discomfort but also prevents procedure-related complications [16]. However, in real practice, maintaining stable levels of sedation or cooperation until the end of the procedure is not easy, especially in cases demanding a long procedure time. There are several reasons for patients’ movement during the ESD procedure. First, a painful stimulation and discomfort during the course of the procedure [8]. In ESD, endoscopic knives directly dissect the submucosal layer and can cause “burn effect” on the muscularis propria, and this process might be accompanied by pain sensation, which can lead to unpredictable patients’ movement. Therefore, administration of analgesics such as pethidine is often effective in patients exhibiting sudden body movement, demonstrating the importance of pain control during the ESD, even under deep sedation [12]. Second, inadequate sedation owing to improper dosage of sedatives. Predicting the dosage of sedatives in each patient before starting the procedure is difficult; therefore, the titration of sedatives requires delicate attention during ESD with dose adjustment considering the patient’s age, sex, medical history, and previous response to sedatives. Consequently, oversedation or undersedation frequently occurs, and patients in undersedation could show some agitation during the procedure [10]. Third, paradoxical response to sedatives; this is the most important problem, which could negatively impact the procedure. History of alcohol abuse, chronic use of narcotics or sleeping pills, genetic predisposition, and psychological background may induce paradoxical body movement, despite proper administration of sedatives [11,17-20]. The incidence of paradoxical reactions following MDZ administration has varied from 1 to 24 % [21-24]. History taking on behavioral responses to previous exposure to sedatives or checking the responses to usual administration of MDZ in pre-ESD endoscopy can help predict the patients’ paradoxical reactions during the ESD procedure.
Patients’ cooperation is not merely the problem of sedation, especially in patients showing paradoxical reactions to sedatives. Therefore, we hypothesized that fixing the patient’s position including the arms to suppress sudden or severe body movement would be helpful in gastric ESD. The focus was on the efficacy of a patient-positioning device on inevitable patient movement despite appropriate sedation, and EZ-FIX® was useful as expected for patients with a higher level of body movement or patients with large lesion size demanding a longer procedure time. In fact, endoscopist satisfaction on sedation and overall satisfaction on the procedure did not show significant difference between two groups. However, EZ-FIX group needed significantly larger amount of propofol and a longer procedure time than the non-EZ FIX, which suggested that more difficult cases including uncooperated patients might have been included in the EZ-FIX group. Therefore, we believe that endoscopist satisfaction profiles might be even lower in the absence of the EZ-FIX® in the EZ-FIX group. In the same vein, although endoscopist satisfaction on sedation and overall satisfaction on the procedure were significantly lower in the contribution group than in the non-contribution group, such results are considered merely a consequence of difficult conditions from large tumor size with a prolonged procedure time and low level of patients’ cooperation, rather than the adverse effects of EZ-FIX® on the procedure. And EZ-FIX® did not cause any procedure-related complications during ESD and showed sufficient satisfaction and safety profiles for patients based on the questionnaire survey. EZ-FIX® is a transformable patient-positioning device with reusable design for a long term (several years) to make the procedure more efficient [25]. It contains polystyrene balls in a soft and non-slip polyurethane cover, and a simple inflation with air and deflation with a vacuum facilitates rapid changes and fixation of the posture of the patient. The patients are more comfortable under a desired posture, and the device also has benefits on inhibiting unpredictable sudden patients’ movement. Additionally, EZ-FIX® might help maintain the patient’s body temperature during the procedure.
In 2015, Lee et al. [25] reported the efficacy and safety of EZ-FIX® in patients undergoing ERCP and described that EZ-FIX® reduced the total dose of propofol and increased the satisfaction of the endoscopists and nurses. To the best of our knowledge, this is the first randomized prospective trial to prove the efficacy of EZ-FIX® on gastric ESD, and benefits of EZ-FIX® were also found in certain circumstances.
Our study has several limitations. First, as a double-blind design was not possible in this study, this might have caused bias in measuring the subjective parameters including the endoscopist satisfaction profiles and the level of contribution of the device. Since there are numerous and even elusive factors affecting the outcomes of gastric ESD including tumors’ characteristics, technical difficulty itself with endoscopist experience, patients’ cooperation with sedation status, and even patients’ respiration and heartbeat, which is difficult to control [26-28], interpreting the subjective variables on the efficacy and contribution of EZ-FIX itself for the procedure was a hard work. However, in our study, for more accurate and unbiased results, two endoscopists together defined parameters with discussions and performed a preliminary test to keep the balanced estimation and finally completed the scores independently. Second, the sample size is relatively small. A large multicenter trial may be beneficial for elucidating the clear role of EZ-FIX. Lastly, anesthesiologists-providing conscious sedation or general anesthesia might be better in patients with a difficult lesion or showing paradoxical reaction, which could make EZ-FIX ineffective. However, in Korea, gastric ESD is still being achieved by an endoscopist, not by an anesthesiologist for the most part, and this means that EZ-FIX might have a role for non-anethesiologists-providing sedation.
In conclusion, EZ-FIX® might be useful especially for patients who are expected to exhibit uncooperative responses to sedatives or patients with large lesion size anticipating a longer procedure time.
Notes
No potential conflict of interest relevant to this article was reported.